There are growing concerns about global warming and the depletion of petroleum resources. Thus, developing renewable energy production and storage technolgies is one of the magjor scientific challenges in our world. The oxygen reduction reaction (ORR) and its reverse reaction, oxygen evolution reaction (OER), are amongst the most studied electrochemical reactions for fundamental reasons as prototypes of four electron
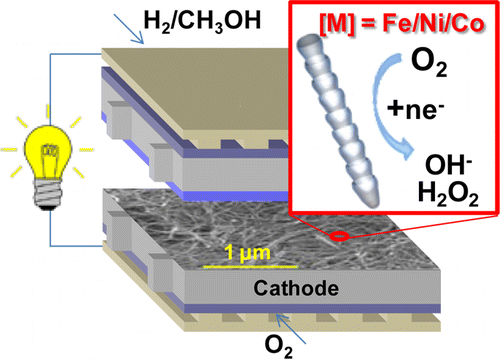
transfer reactions, and for technological relevance in renewable energy production. Specifically, both ORR and OER are at the center of many applications in electrochemical energy conversion processes including polymer electrolyte membrane fuel cells (PEMFCs), direct-solar and electrolytic water-splitting devices, and metal-air batteries. However, a key element for the commercialization of these reactions is the need of an efficient and cost-effective catalyst that solves their slow kinetics at the oxygen electrodes, namely the cathode in fuel cells and the anode in electrolyzers. For example, Pt-based alloys are among the best catalysts in PEMFCs despite the fact that the fuel cells have a significant power loss due to a low operating voltage of 0.7 V as measured with respect to standard hydrogen electrode (SHE); this is only 57% of the available free energy. Additionally, and more importantly, large amounts of the precious metal Pt are needed to boost the cathode kinetics that are significantly slower than the hydrogen evolution at the anode.
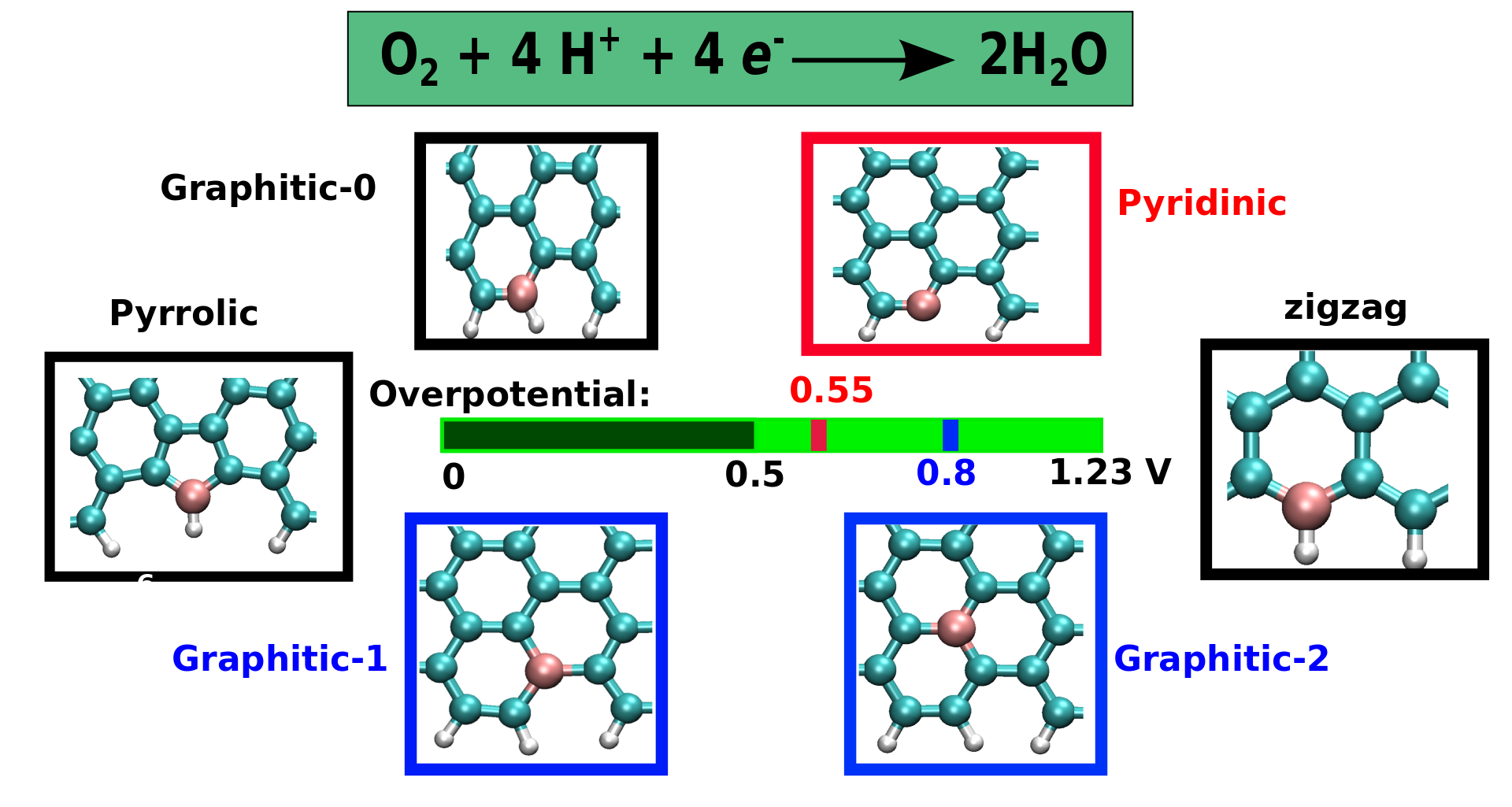
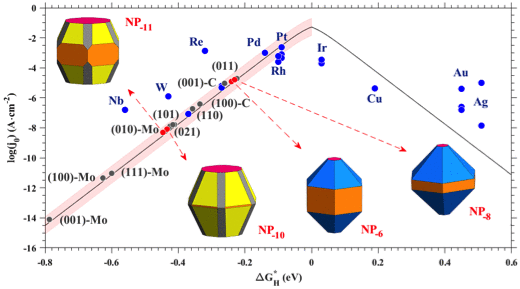
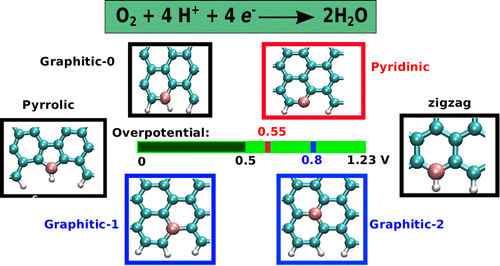
In our recent study, we investigated using first-principles density functional theory the electrocatalytic activity towards the four-electron oxygen reduction-reaction in N-doped graphene quantum dots.